BOUTIQUEResources
In the field of innovative drug
development, particularly for high-concentration protein formulations, the
solubility and long-term stability of the Active Pharmaceutical Ingredient
(API) remain key bottlenecks determining drug development success. With the
biopharmaceutical industry's growing demand for high-concentration
formulations, the challenges of protein aggregation and precipitation under
high-concentration conditions have become increasingly prominent. Pfanstiehl's
newly launched novel pharmaceutical excipient, A-172 (Arginine Glutamate), is
specifically designed to address these escalating formulation challenges in
biologic drug development.
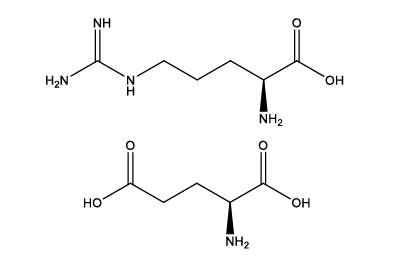
A-172 is formed through ionic bonding between two natural amino acids found in the human body—arginine and glutamate—creating a structurally stable amino acid ion-pair salt. It effectively addresses common issues in high-concentration protein formulations, including low solubility, susceptibility to aggregation, and poor stability. Its zwitterionic properties enable effective interaction with protein surfaces, enhancing hydrogen bonding and electrostatic interactions while forming stable ion pairs. This significantly improves protein drug solubility and inhibits aggregation, thereby ensuring the stability of high-concentration formulations. Since its constituent amino acids are endogenous to the human body, A-172 demonstrates lower toxicity and superior biocompatibility, making it particularly suitable for subcutaneous injection formulations that require frequent administration and high patient compliance.
Why A-172 (Arginine Glutamate) is an Excellent Excipient for Injectable Biologic Formulations:
1. Reducing Formulation Viscosity
A-172 helps regulate the osmotic pressure of injectable solutions to physiological levels, improving the rheological properties of the drug solution, reducing viscosity, and enhancing flowability. This characteristic is particularly suitable for sensitive administration routes such as subcutaneous injection, reducing injection pain and tissue damage while improving the manufacturability of the formulation.
2. Enhancing Protein Solubility
A-172 significantly improves the solubility of poorly soluble drugs in aqueous systems, ensuring effective drug release and bioavailability in vivo. It demonstrates excellent compatibility with peptides, proteins, antibodies, and small-molecule chemical drugs, providing an ideal dissolution environment for protein-based therapeutics.
3. Reducing Protein Aggregation
By stabilizing protein structure, A-172 significantly reduces aggregation phenomena in high-concentration protein formulations, ensuring biologic drug activity and maintaining efficacy and safety during storage and administration. This is critical for preserving the quality stability of biologic formulations.
4. Minimizing Liquid-Liquid Phase Separation
The natural buffering capacity of A-172 maintains stable pH in injectable solutions, preventing phase separation caused by pH drift. Its broad drug compatibility helps maintain formulation homogeneity, ensuring the product remains in a stable physical state throughout its storage period.
5. Improving Thermal Stability
Through multiple stabilization mechanisms including inhibition of oxidation, aggregation, and pH drift, A-172 significantly enhances the physicochemical stability of injectable formulations, effectively extending product shelf life and providing reliable protection under various storage conditions.
Pfanstiehl, maintaining its unwavering commitment to premium quality standards, is pleased to introduce injectable-grade L-Arginine L-Glutamate (A-172). This product delivers ultra-high purity, ultra-low endotoxin levels, and minimal metal ion content. Comprehensive quality testing covers DNA, RNA, DNase, RNase, phosphatase, protease, lipase, nitrite, mycoplasma, and additional critical parameters. A-172 is suitable for both biopharmaceutical and small-molecule drug manufacturing, with exceptional applicability for injectable formulation development.
Product Features:
Ultra-high purity, ultra-low endotoxin, and ultra-low microbial content
- Low metal impurity residue: 29 metal ions individually tested, all at ppb levels
- Tested for DNA, RNA, DNase, RNase, phosphatase, protease, lipase, and mycoplasma residues
- Tested for nitrite residues
- Tested for β-Glucans residues
- Manufactured in strict compliance with ICH-Q7 cGMP standards
- Multiple packaging specifications to support flexible applications from R&D to commercial production
Product Applications:
- pH Buffering Agent
- Viscosity-Reducing Agent
- Stabilizer
- Solubility Enhancer
Product Information:
|
Product Name |
CAS Number |
Product Code |
Pharmacopoeia Compliance |
DMF |
CDE |
CDE Status |
|
L-Arginine L-Glutamate,High Purity,Low Endotoxin, Low Metals GMP |
4320-30-3 |
A-172 |
Pfanstiehl sponsoring USP/NF monograph |
42345 |
F20250000567 |
I |


 京公網安備 11010802028692號
京公網安備 11010802028692號