BOUTIQUEResources
In the manufacturing of biologics, residual host cell DNA (HCD) may persist, potentially introducing oncogenes or other risky genetic materials into the final drug product. To mitigate these risks, regulatory agencies have established limits for HCD residues. Depending on the cell line and dosing regimen, allowable HCD limits typically range from 10 to 100 pg per dose. Regarding HCD testing, the Chinese Pharmacopoeia (ChP) 2025, General Chapter 3407, outlines three methods: DNA probe hybridization, fluorescence staining, and quantitative PCR (qPCR). Meanwhile, USP General Chapter <509> "Residual DNA Testing" recommends probe-based DNA quantification as a validated method for testing recombinant biologics produced in E. coli or Chinese Hamster Ovary (CHO) cell lines to ensure superior sensitivity and accuracy. Throughout the production process, Quality Control (QC) personnel must evaluate DNA levels in samples that may contain other impurities and high concentrations of active pharmaceutical ingredients (API), necessitating a reliable and sensitive DNA quantification method.
The 2025
edition of the ChP has introduced new HCD testing requirements for several
monoclonal antibodies, including Trastuzumab for injection, Infliximab,
Adalimumab, Bevacizumab, and Rituximab. It also specifies requirements for
certain vaccines (e.g., inactivated Sabin strain poliovirus vaccine <50 pg/dose), explicitly mandating the use of General Chapter 3407 Method 3 (qPCR). Additionally, a new general chapter,
The
newly launched Cygnus AccuRes? series of DNA
quantification kits is developed in accordance with ChP General Chapter 3407
Method 3 (qPCR). These kits are specifically designed for HCD testing in
biologics recombinantly expressed in cell lines such as CHO, Human, Vero, and
E. coli. High-concentration samples can be tested with minimal dilution,
effectively lowering the Limit of Detection (LOD).

Key Advantages of Cygnus AccuRes? HCD
Detection Kits:
- Accuracy: Cygnus’s proprietary DNA extraction reagents remove components that interfere with PCR. The probe-based method ensures highly specific detection of target cell lines/species (CHO, Human, Vero, E. coli), avoiding interference from heterologous DNA. FAM-labeled nucleic acid probes are quenched by BHQ-1? prior to PCR amplification to ensure specificity.
- Enhanced Precision: The CleanAmp® dNTPs and Hot-Start Taq DNA Polymerase system effectively reduce primer-dimers and non-specific amplification before the PCR reaction reaches the denaturation temperature.
- Flexibility: Compatible with all qPCR instruments capable of detecting FAM signals, reducing the need for additional equipment investment.
- All-in-One Kits: Supports the entire workflow from sample preparation to qPCR. Kits include DNA extraction reagents (tube or plate format), AccuRes? PCR Master Mix, Primer/Probe Mix, and DNA standards.
- Sensitivity: LODs are 0.6 fg/μL (CHO), 0.7 fg/μL (E. coli), and 3 fg/μL (Human).
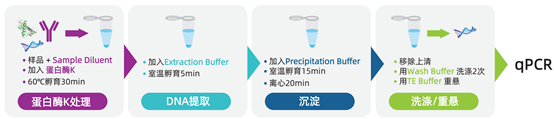
Workflow of Cygnus AccuRes? HCD Detection Kits
1. DNA Extraction: Cygnus utilizes a novel DNA carrier capable of recovering femtogram levels of HCD. The extraction process occurs in an environment free from protein, salt, and surfactant contamination, resulting in higher reproducibility and stability for DNA detection and amplification compared to other methods.
2. Amplification: The kits use a highly specific primer/probe system. FAM-labeled probes are quenched by BHQ-1? in every PCR cycle. The advanced CleanAmp® dNTPs and Hot-Start Taq system ensure specificity, sensitivity, and stability, while allowing qPCR setup at room temperature.
3. Quantification: A standard curve is constructed using the Ct values of kit standards, calculating results as pg/10 μL of residual host cell DNA. This can be converted to ng/mL, ng/mg, or ng/dose. Based on this method, the Lower Limit of Quantification (LLOQ) is 0.6 fg/μL for CHO.
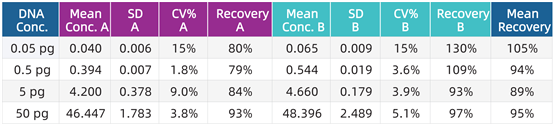
Test Data Summary
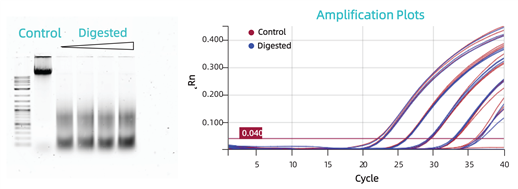
Impact of DNA Degradation: Comparison of extraction and amplification between intact gDNA
(Control) and CHO DNA digested with Alu1 enzyme for 30, 60, 90, and 120
minutes. The linear range of the Control and the 30-minute digested sample
remained consistent.
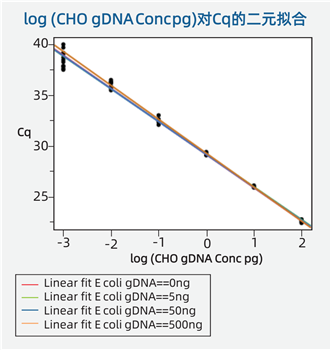
Specificity: The CHO primer/probe system maintains a wide linear range and high
specificity even in the presence of exogenous DNA (e.g., various concentrations
of E. coli DNA do not affect the CHO DNA standard curve).
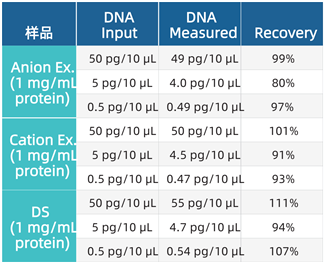
Process Samples & Drug Substance
(DS): Recovery of CHO DNA was compared in samples
containing 1 mg/mL of protein purified via anion or cation exchange, as well as
in final drug substances.
Ordering Information
|
Product Name |
Catalog No. |
Size |
|
CHO AccuRes? DNA Quantification Kit in Tubes |
D1555T |
1 kit |
|
CHO AccuRes? DNA Quantification Kit in Wells |
D1555W |
1 kit |
|
CHO AccuRes? Quantitative DNA Kit |
D1555 |
1 kit |
|
E. coli AccuRes? Quantitative DNA Kit |
D1415 |
1 kit |
|
Human AccuRes? Quantitative DNA Kit |
D1165 |
1 kit |
|
Vero AccuRes? Quantitative DNA Kit |
D1975 |
1 kit |
|
NS/0 AccuRes? Quantitative DNA Kit |
D1225 |
1 kit |
Upcoming Products:
|
Product Name |
Catalog No. |
Size |
|
SF9 AccuRes? Quantitative DNA Kit |
D1845 |
1 kit |
|
P. pastoris AccuRes? Quantitative DNA Kit |
D1145 |
1 kit |
Cygnus DNA Extraction Kits
These kits are designed to be used in conjunction with the AccuRes? quantification kits.
|
Product Name |
Catalog No. |
Size |
|
DNA Extraction Kit in Tubes |
D100T |
1 kit |
|
DNA Extraction Kit in Wells |
D100W |
1 kit |


XMJ Scientific (Beijing) Co., Ltd. As the exclusive distributor for Cygnus in China, XMJ Scientific has established long-term and stable partnerships with numerous well-known domestic pharmaceutical companies and CRO/CMO enterprises. For years, XMJ’s products and services have helped many companies accelerate R&D, improve drug quality, purity, and safety, optimize manufacturing processes, reduce time-to-market, and lower QC costs


.png) 京公網(wǎng)安備 11010802028692號(hào)
京公網(wǎng)安備 11010802028692號(hào)